
Introduction
Polymers are inherently vulnerable to oxidative degradation during high-temperature processing and long-term service. Without adequate protection, they lose mechanical strength, colour, and durability prematurely — compromising product performance and lifespan.
The data shows how quickly damage accumulates. A polypropylene reprocessing study found that melt flow rate increased by approximately 470% after just 5 recycling cycles at 230°C, a direct consequence of oxidative chain scission. The presence of oxygen also reduces PP's thermal decomposition onset temperature by roughly 75°C compared to inert atmospheres — a critical concern during extrusion and injection moulding.
This guide breaks down how antioxidant additives work, which types to use for different polymers and processing conditions, and why they're becoming essential in closed-loop recycling systems.
TL;DR
- Oxidation in polymers — triggered by heat, oxygen, and UV — causes discoloration, embrittlement, and mechanical property loss
- Primary antioxidants neutralize free radicals; secondary antioxidants decompose hydroperoxides before chain degradation accelerates
- Main types are phenolic, phosphite/phosphonite, and thioester antioxidants — synergistic blends outperform single-type systems
- Selection depends on polymer type, processing temperature, end-use environment, and regulatory requirements (FDA, EU 10/2011)
- Essential across packaging, automotive, agricultural film, and recycling — anywhere long-term thermal stability is non-negotiable
Why Polymers Degrade: The Role of Oxidation
The Auto-Oxidation Mechanism
Polymer auto-oxidation follows the Bolland-Gee radical chain mechanism, proceeding through four stages:
- Initiation: Formation of the first radicals (R•) at weak points in the polymer chain
- Propagation: R• reacts with oxygen to form peroxy radicals (ROO•), which then abstract hydrogen from adjacent polymer chains to form hydroperoxides (ROOH) while generating new radicals
- Chain branching: Hydroperoxides decompose to produce additional radicals, accelerating the cycle
- Termination: Radical recombination halts the process temporarily
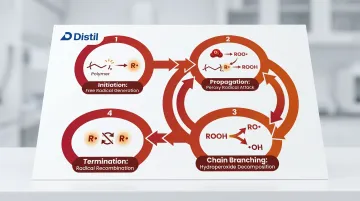
In polypropylene, the tertiary hydrogen is the most vulnerable site for radical attack. Studies show that oxidation is "infectious": it initiates at specific sites (such as catalyst residues) and spreads heterogeneously through the polymer matrix rather than occurring uniformly.
Measurable Degradation Consequences
Melt Flow Rate Increase: Virgin PP exhibits a melt flow rate of 10-12 g/10 min at standard test conditions. After 5 reprocessing cycles, MFR increased by 470% (at 230°C), attributed to molecular weight reduction via chain scission — a clear indicator of severe oxidative damage.
Thermal Stability Loss: Virgin PP's decomposition onset in air occurs at 264°C versus 339°C in nitrogen — approximately 75°C lower due to oxygen presence. After 5 reprocessing cycles, this onset drops further to 239°C, demonstrating cumulative oxidative vulnerability.
Mechanical Property Decline: Elongation at break (EAB) initially increases during early aging stages due to chemi-crystallization, then declines substantially. At 125°C aging, EAB peaked at 29 days before declining significantly by 84-98 days. Molecular weight (Mn) decreased progressively with both aging time and temperature.
Processing Stages and Oxidative Risk
These cumulative degradation effects are most acute during melt processing. At 180-230°C in air, thermal decomposition dominates over mechanical shear during extrusion and injection molding. The activation energy for oxidative degradation starts at approximately 70 kJ/mol during early stages (peroxide radical decomposition) and rises to approximately 160 kJ/mol at later stages (random chain scission).
Processing is the highest-risk window, but it is not the only one. Long-term service-life degradation in oxygen-rich environments — particularly in outdoor applications exposed to heat and UV — poses compounding risks that shorten service life.
Types of Antioxidant Additives for Polymers
Antioxidant additives are classified into primary and secondary antioxidants based on their mechanism of action, with thioester antioxidants forming a third category. Each addresses a different stage of the oxidation chain, and synergistic combinations significantly outperform single-type systems.
Primary Antioxidants: Hindered Phenols
Hindered phenolic antioxidants donate hydrogen atoms to peroxy radicals (ROO•), terminating the chain reaction. The resulting phenolic radical is stabilized by resonance and steric hindrance from bulky tert-butyl groups. This makes them the go-to choice for long-term thermal stability.
Common hindered phenol stabilizers include:
- Irganox 1010 (Pentaerythritol tetrakis(3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate)): Molecular weight 1178 g/mol, melting range 110-125°C, flash point 297°C. Described as a highly effective, non-discoloring, sterically hindered primary antioxidant suitable for polyolefins (PE, PP), PET, and engineering plastics.
- Irganox 1076 (Octadecyl 3,5-di-tert-butyl-4-hydroxyhydrocinnamate): Noted for odorless performance, high extraction resistance, and excellent color retention — preferred for food packaging applications.
Recommended loading for polyolefins: 0.05-0.4% by weight.
Secondary Antioxidants: Phosphites and Phosphonites
Phosphite and phosphonite antioxidants decompose hydroperoxides (ROOH) into stable, non-radical alcohols before they can generate new radicals during the propagation phase. The phosphite is oxidized to a phosphate in the process — making this class most active during high-temperature melt processing.
The most widely used phosphite stabilizer:
- Irgafos 168 (Tris(2,4-di-tert-butylphenyl)phosphite): A hydrolytically stable phosphite processing stabilizer that reacts with hydroperoxides formed during autoxidation, preventing process-induced degradation and extending the performance of primary antioxidants.
Research confirms that phosphites protect both the polymer and phenolic antioxidants during melt processing, reducing hydroperoxide levels while preserving the effectiveness of primary stabilizers.
Typical loading: 0.05-0.2% by weight, often in combination with phenolics.
Thioester / Sulfur-Based Antioxidants
Thioester antioxidants such as DSTDP (distearyl thiodipropionate) and DLTDP (dilauryl thiodipropionate) decompose hydroperoxides through a series of oxidation reactions. Unlike phosphites, which excel during high-temperature processing, thioesters function primarily at lower temperatures over extended periods — making them ideal for long-term heat aging stability in polyolefins.
Temperature-dependent crossover effects are worth noting here. At 150°C, Irganox 1010 + DSTDP combinations outperform alternative phenolic + thioester blends. At 80°C, the reverse holds — which is why matching antioxidant chemistry to service temperature matters as much as the selection itself.
Why Synergistic Blends Are Standard Practice
Research on stabilizer combinations confirms that ternary blends of primary, secondary, and thioester antioxidants significantly outperform single-type systems. Phosphites reduce consumption of phenolic antioxidants during processing by eliminating hydroperoxide precursors. Thioesters extend long-term heat aging resistance. Together, they cover both high-temperature processing and extended service life.
Commercial antioxidant packages often combine:
- Phenolic (e.g., Irganox 1010) at 500-1000 ppm
- Phosphite (e.g., Irgafos 168) at 500-1500 ppm
- Thioester (e.g., DSTDP) at 300-500 ppm

How to Select the Right Antioxidant Additive for Your Application
Polymer Matrix Compatibility
Different polymer backbones exhibit varying sensitivities and compatibility requirements. The table below maps common polymer types to their appropriate antioxidant strategies:
| Polymer | Oxidation Risk | Recommended Antioxidant Type | Typical Loading |
|---|---|---|---|
| Polyolefins (PE, PP) | High — tertiary carbon sites | Hindered phenolics (Irganox 1010/1076) + phosphites (Irgafos 168) + thioesters | 0.1–0.4% |
| PET | High at processing temps >280°C | High-MW phenolics (>1000 g/mol), low vapor pressure | 0.05–0.3% |
| Engineering Plastics (Nylon, PC) | Severe above 280°C; polyamide-specific degradation pathways | Phenolics with amide linkages — resist volatilization and stabilize melt | 0.05–0.3% |
Processing Temperature Window
Match antioxidant volatility and thermal stability to the polymer's processing temperature:
- Below 230°C (PE, PP): Standard hindered phenolics and phosphites perform well
- 230–280°C (PET, nylon): Require high-MW antioxidants (>1000 g/mol) with low volatility
- Above 280°C (polycarbonate, high-performance engineering plastics): Demand antioxidants with flash points exceeding 290°C and vapor pressures below 1×10⁻⁹ Pa
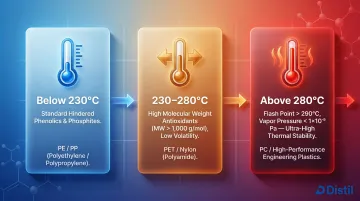
Irganox 1010, with a flash point of 297°C and vapor pressure of 7×10⁻¹⁰ Pa at 20°C, is a benchmark for processing-stable antioxidants in high-temperature applications.
Processing temperature selection directly shapes which regulatory frameworks apply — particularly for food-contact and medical-use polymers.
End-Use and Regulatory Requirements
FDA 21 CFR 178.2010: Governs antioxidant additives permitted in food-contact polymers. Approved substances include Irganox 1010, Irganox 1076, Irgafos 168, and thiodipropionic acid esters, with concentration limits typically ranging from 0.1–0.5% by weight. Usage must not exceed the amount reasonably required for the intended technical effect.
EU Regulation 10/2011: Establishes specific migration limits (SML) for authorized substances:
| Antioxidant | SML (mg/kg food) |
|---|---|
| Irganox 1076 | 6 |
| Irgafos 168 | 10 |
| Irganox 3114 | 5 |
| DLTDP | 5 |
| Default (unlisted) | 60 |
Actual migration testing in biodegradable tableware found Irganox 1010 migrated at 2.77 mg/kg maximum (in 95% ethanol) and Irgafos 168 at 4.23 mg/kg — well below regulatory limits.
REACH Compliance: Irganox 1010 does not contain substances at levels ≥0.1% listed in the REACH Authorization or Candidate Lists, confirming that widely used polymer antioxidants are not currently classified as Substances of Very High Concern (SVHCs) under European regulations.
Dosage and Loading Levels
Typical concentration ranges by polymer:
- Polyolefins: 0.05–0.4%
- Hot melt adhesives: 0.2–1%
- Engineering plastics: 0.05–0.3%
- General long-term thermal stability: 0.05–0.1%
Consequences of improper dosing:
- Under-dosing: Protection depletes before the product reaches end-of-life, accelerating embrittlement and discoloration in service
- Over-dosing: Exceeding solubility limits causes blooming — phase separation and surface migration — which creates aesthetic defects and reduces surface degradation resistance. Studies specifically flag Irganox 1076 and Irganox 3114 as prone to blooming on polyurethane and medical-grade elastomers.
Distil's polymer additives team works with packaging converters, automotive suppliers, and healthcare compounders to develop application-specific antioxidant packages — including compatibility trials, dosage optimization, and regulatory documentation — from lab scale through commercial production.
Antioxidant Additives by Industry Application
Packaging (Flexible Films, Rigid Containers)
Challenge: Packaging converters face dual requirements of food safety compliance and processing stability in PE/PP films and PET bottles. Antioxidants must ensure clarity retention and avoid affecting taste or odor of packaged food.
Solution: A BASF case study on LLDPE food packaging film demonstrated that replacing 50% of Irgafos 168 (1000 ppm → 500 ppm) with 100 ppm Vitamin E (Irganox E 201) reduced NIAS migration of 2,4-di-tert-butylphenol (a phosphite hydrolysis product) by approximately 75% — from ~240 µg/kg to ~60 µg/kg. Melt flow stability was maintained across 5 extrusion passes at 230°C.
Irganox 1076 is the preferred phenolic antioxidant for food packaging due to its odorless performance, high extraction resistance, and excellent color retention.
Automotive and Wire & Cable
Automotive Under-the-Hood Components: High-heat, long-service-life demands require antioxidants that perform reliably over decades. Components are exposed to continuous thermal cycling (80–150°C), mechanical stress, and oxygen exposure.
Wire and Cable Insulation: Oxidation Induction Time (OIT) testing at 200°C per IEC 60811 is the standard evaluation method for antioxidant effectiveness in XLPE insulation. Key performance factors include:
- Synergistic blends: Compound formulations (such as Antioxidant 300 + Antioxidant 1081 at 4:1 ratio) deliver higher OIT than single-component systems
- Crosslink density: Higher crosslinking reduces antioxidant migration and volatilization
- Oxygen barrier effect: Denser crosslinked networks slow oxygen penetration, extending service life
Distil formulates masterbatch systems for automotive and wire & cable applications, with R&D-led optimization for thermal stability, flame retardancy, and impact strength across demanding under-the-hood and insulation environments.
Agriculture and Construction
Outdoor Service Life Requirements: Agricultural films (mulch, greenhouse covers) and construction piping face combined UV and thermal stress over extended periods.
Service life data for agricultural PE films:
- Standard greenhouse films: 1 year / 2 seasons
- Long-life greenhouse films: 2–3 years (up to 33 months)
- Anti-fog monolayer films: 6–12 months
Service life is defined as the point where residual elongation at break is at least 50% of original value.
Integrated Additive Packages: Effective outdoor stabilization requires combining HALS (hindered amine light stabilizers), UV absorbers, and antioxidants. For environments with agrochemical exposure (sulfur, chlorine), NOR HALS + antioxidant combinations provide resistance to up to 3000 ppm sulfur while maintaining 2–3 year service life.
Distil's agricultural film additive packages are developed with application-specific trials — covering UV stabilization, anti-fog performance, and agrochemical resistance — to match the actual service conditions of each film type.
Antioxidants in Polymer Recycling
The Recycling Challenge
Each mechanical recycling cycle exposes the polymer to further thermal oxidation during remelting and reprocessing, progressively depleting residual antioxidant reserves. This leads to quality degradation in recycled resins (rPET, rPE, rPP) and limits the number of viable recycling loops.
Antioxidant Depletion and Build-Up
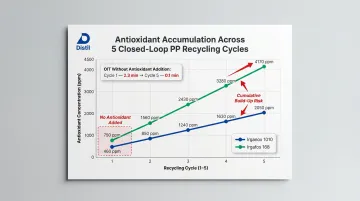
Research monitoring antioxidant consumption across 5 closed-loop recycling cycles for PP (adding 500 ppm Irganox 1010 and 1000 ppm Irgafos 168 per cycle) revealed:
| Cycle | Irganox 1010 (ppm) | Irgafos 168 (ppm) | OIT at 225°C (min) |
|---|---|---|---|
| 1 | 460 | 750 | 2.9 |
| 2 | 850 | 2180 | — |
| 5 | 2050 | 4170 | 36.6 |
| 5 (no AO) | — | — | 0.1 |
Key findings:
- Irgafos 168 (phosphite) depletes more rapidly than Irganox 1010 (phenolic) during initial processing
- Significant cumulative build-up occurs in closed-loop systems if standard doses are added every cycle — reaching over 4× initial loading
- Without antioxidant addition, OIT dropped from 2.3 min to 0.1 min by cycle 5, indicating near-complete loss of oxidative stability
Restabilization Strategies
Three approaches have demonstrated practical value for maintaining polymer quality through recycling:
- Add fresh antioxidants directly to recycled material rather than diluting with virgin polymer. Research found that adding up to 80% virgin polymer to degraded PP had "no effect" on improving recyclability — oxidation spreading from degraded zones cannot be stopped by dilution alone.
- Select higher-MW stabilisers (such as Irganox 1010) for virgin PP from the outset. These resist volatilisation better than lower-MW alternatives, providing a stronger baseline for repeated recycling. Notably, in-use degradation during service life has a larger negative impact on recyclability than multiple melt-processing steps.
- **Calibrate dosing with OIT testing** rather than applying fixed restabilisation doses per cycle. DSC-based Oxidation Induction Time (OIT) measurement per ASTM D3895 / ISO 11357-6 allows recyclers to assess residual antioxidant capacity and dose only what is needed — preventing the cumulative build-up shown in the table above.
Polymeric/Bound Antioxidants for PET Recycling
The PP-focused strategies above address restabilisation through dosing. For PET, the challenge is different: conventional low-MW antioxidants volatilise and migrate at PET's high processing temperatures (above 280°C), making standard restabilisation less effective.
Research published in Macromolecules addresses this by incorporating antioxidant moieties into a polymeric backbone or high-MW structure. These polymeric additives resist volatilisation and migration, maintaining molecular weight, reducing yellowing, and preserving mechanical properties across multiple recycling cycles. The additive topology — such as multi-arm or star-like structures — plays a critical role in effectiveness, representing an emerging approach for closing the PET recycling loop.
Frequently Asked Questions
What are antioxidant additives?
Antioxidant additives are chemical stabilizers incorporated into polymers to inhibit or prevent oxidative degradation caused by exposure to heat, oxygen, and UV radiation during processing or service life. They function by interrupting the auto-oxidation chain reaction at different stages.
What are antioxidant plastic additives used for?
Antioxidant additives serve several critical functions in polymer systems:
- Prevent discoloration and yellowing during processing and service
- Maintain mechanical properties such as tensile strength and elongation at break
- Extend service life in oxygen-rich environments
- Ensure processing stability during extrusion, injection molding, and other high-temperature operations
What are the antioxidants used in polymers?
The main categories are hindered phenolic antioxidants (primary, such as Irganox 1010 and 1076), phosphite/phosphonite antioxidants (secondary, such as Irgafos 168), and thioester antioxidants (such as DSTDP and DLTDP). These are often used in synergistic blends tailored to the specific polymer and application requirements.
What is the difference between primary and secondary antioxidants in polymers?
Primary antioxidants (hindered phenolics) interrupt the free radical chain reaction by donating hydrogen atoms to peroxy radicals, terminating propagation. Secondary antioxidants (phosphites, thioesters) decompose hydroperoxides before they can generate new radicals — addressing degradation at an earlier stage. Used together in synergistic blends, they cover both the initiation and propagation phases of oxidation.
How do you select the right antioxidant additive for a polymer application?
Key selection factors include:
- Polymer type: polyolefin, PET, or engineering plastic determines base compatibility
- Processing temperature: must match antioxidant volatility and thermal stability range
- End-use conditions: outdoor vs. indoor exposure, thermal cycling, and target service life
- Regulatory requirements: food contact applications require FDA 21 CFR or EU 10/2011 compliance
Application-specific formulation support is recommended to balance these variables effectively.
Are antioxidant additives safe for food contact packaging?
Many antioxidant additives are approved for food contact use under FDA 21 CFR 178.2010 and EU Regulation 10/2011, but compliance must be verified for specific additive grades, dosage levels, and intended food-contact applications. Migration testing against substance-specific SMLs is required for regulatory approval.
Need application-specific antioxidant formulation support? Distil's polymer additives team supports the full process — from compatibility trials and dosage optimization to regulatory alignment and scale-up. Contact us at +91 7902101101 or contact@distil.market.



