
Introduction
Most commodity plastics — polyethylene, polypropylene, polystyrene, and polyurethane — share a critical vulnerability: they're inherently combustible. When exposed to fire, these hydrocarbon-rich polymers decompose into flammable gases, release dense smoke and toxic compounds, and generate extreme heat. For manufacturers serving automotive, wire and cable, electronics, and construction markets, this isn't just a technical concern — it's a regulatory and safety imperative.
Flame retardant (FR) additives are engineering tools designed to disrupt the combustion cycle, preventing ignition, slowing flame spread, or reducing heat release enough to meet strict fire safety standards. Unlike coatings or surface treatments, FR additives are integrated directly into the polymer matrix during compounding, delivering consistent performance across the material's lifecycle.
Getting FR selection right — mechanism, chemistry, and regulatory fit — determines whether a formulation passes certification or fails on the production floor. What follows breaks down each of those decisions.
TL;DR
- Polymers burn through a self-sustaining cycle of thermal decomposition, fuel release, and combustion that FR additives disrupt
- Three mechanisms dominate: gas-phase radical inhibition, endothermic cooling, and condensed-phase char formation
- Key FR classes: halogenated, phosphorus-based, mineral fillers, intumescent systems, and synergists
- Reactive FRs bond into the polymer backbone, eliminating migration but requiring custom synthesis
- Selection depends on polymer type, processing temperature, target fire test standard, and REACH/RoHS constraints
How Polymers Burn: Understanding the Combustion Cycle
Polymer combustion is a self-reinforcing feedback loop with four distinct stages:
- External heat causes thermal decomposition — the solid polymer breaks down into smaller molecular fragments
- Molten or gaseous fuel migrates to the flame front — these fragments volatilize and move toward the combustion zone
- Flammable gases combust with oxygen — generating heat, light, and radiant energy
- Radiant energy feeds back to the polymer — sustaining pyrolysis and creating a continuous cycle
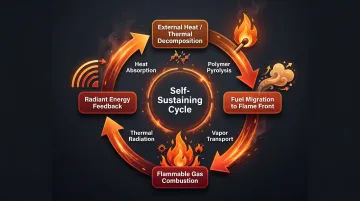
Without intervention, this cycle accelerates until the fuel source is exhausted or oxygen is depleted. How fast it escalates — and where it's most vulnerable to disruption — depends heavily on the polymer's own chemistry.
Polymer Chemistry Determines Fire Behavior
Chemical structure directly governs flammability. The cone calorimeter, developed by NIST and standardized under ISO 5660-1, measures heat release rate (HRR) — the most critical metric for fire hazard assessment.
| Polymer | Peak HRR (kW/m²) | Characteristics |
|---|---|---|
| Polyethylene (PE) | 700–1,100 | Pure hydrocarbon; melts, drips; no char |
| Polypropylene (PP) | 800–1,200 | Very high HRR; rapid non-charring burn |
| Polystyrene (PS) | 700–1,300 | High HRR; heavy black smoke |
| PVC | 100–250 | Low HRR; chlorine acts as inherent FR |
| Polycarbonate (PC) | 300–600 | Moderate; forms protective char layer |
Pure hydrocarbon polymers like PE and PP release 5–10× more heat than PVC because they decompose entirely into gaseous fuel without forming a protective char barrier. PVC's chlorine content releases HCl during pyrolysis, which scavenges free radicals in the flame zone and reduces effective combustion.
Polycarbonate behaves differently. It undergoes cyclization during burning, forming a carbonaceous char that physically shields the underlying material — a structural response that cuts peak HRR by more than half compared to polystyrene.
Mechanism-matching is therefore central to effective FR design. An additive that interrupts gas-phase combustion will do little for a polymer that fails through rapid char-free melt dripping. What passes a vertical burn test may still fail a heat release test — because each addresses a different point in the same cycle.
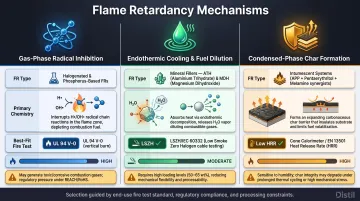
Three Mechanisms of Flame Retardancy
Mechanism 1: Gas-Phase Radical Inhibition
Gas-phase FRs — primarily halogenated and some phosphorus compounds — release reactive species during thermal decomposition that scavenge the H• and OH• free radicals driving combustion.
Brominated FRs release HBr, which reacts with hydroxyl radicals to form water and stable bromine radicals, terminating the chain reaction. Organophosphorus FRs decompose into volatile PO• and PO₂• radicals that capture hydrogen and hydroxyl radicals in the flame zone.
This mechanism functions as chemical quenching of the combustion reaction. At typical loadings of 8–18 wt%, it delivers strong performance per unit weight — but carries a documented trade-off: by inhibiting complete oxidation, gas-phase FRs shift combustion products toward CO and soot rather than CO₂ and H₂O, increasing smoke density and toxicity.
Mechanism 2: Endothermic Cooling and Fuel Dilution
Mineral filler FRs operate through dual condensed-phase and gas-phase action:
- Aluminum trihydrate (ATH) decomposes at 180-200°C, releasing up to 34.6% of its weight as water vapor
- Magnesium hydroxide (MDH) decomposes at 330°C, making it suitable for higher-temperature processing
Both materials absorb heat endothermically, cooling the polymer surface and slowing pyrolysis. Released water vapor dilutes the concentration of flammable gases reaching the flame, reducing the fuel-to-oxygen ratio below the combustion threshold. This mechanism produces a clean combustion profile with minimal smoke or toxic gas generation — the reason ATH and MDH dominate low-smoke zero-halogen (LSZH) cable applications.
That clean profile comes at a cost: meaningful FR performance typically requires 50–60 wt% loading, which significantly impacts mechanical properties like tensile strength and elongation.
Mechanism 3: Condensed-Phase Char Formation
Intumescent systems and nanocomposites form a solid carbonaceous or mineral-rich barrier on the polymer surface when exposed to heat. This char layer:
- Insulates the underlying polymer from radiant heat
- Physically blocks release of flammable decomposition gases
- Slows the mass-loss rate, starving the flame of fuel
Intumescent system chemistry: Three components drive the reaction — an acid source (ammonium polyphosphate), a carbon source (pentaerythritol or the polymer itself), and a blowing agent (melamine). Under fire exposure, the acid catalyzes dehydration of the carbon source into char while the blowing agent expands it into a thick insulating foam.
Synergistic Combinations
Combining mechanisms consistently outperforms single-mechanism systems:
- Phosphorus-nitrogen synergy — phosphorus promotes dehydration while nitrogen-containing compounds increase char density and cohesion; commonly used in intumescent coatings
- Halogen-antimony oxide synergy — antimony trioxide (Sb₂O₃) amplifies brominated FR performance through radical trapping and oxygen sealing in the gas phase; typical ratios range from 1:2 to 1:3.5 (Sb₂O₃:Br)
The mechanism governs fire test outcomes. Gas-phase FRs excel at ignition resistance and flame spread tests (UL 94 V-0), while char-forming systems perform better in structural integrity and heat release tests. The end-use fire standard must drive FR selection from the start.
Types of Flame Retardant Polymer Additives
Halogenated Flame Retardants
Brominated FRs dominate this class, with aromatic brominated compounds widely used in electronics casings, wire and cable jackets, and epoxy circuit boards. Common examples include decabromodiphenyl ether (DecaBDE), tetrabromobisphenol A (TBBPA), and ethylene bis-tetrabromophthalimide (EBTBPI).
How they work: High bromine content per molecule provides potent gas-phase inhibition at relatively low loadings (8-18 wt%). Antimony trioxide (Sb₂O₃) is the standard synergist, used at validated ratios:
- ABS: 4 phr Sb₂O₃ / 14 phr Br (1:3.5 ratio)
- HIPS: 4 phr Sb₂O₃ / 12 phr Br (1:3 ratio)
- PP/PE: 5 phr Sb₂O₃ / 15 phr Br (1:3 ratio)
Regulatory pressure: The European Chemicals Agency (ECHA) identified aromatic brominated FRs as candidates for EU-wide restriction in March 2023. Decabromodiphenyl ethane (DBDPE) was added to the SVHC Candidate List in November 2025, classified as very persistent, very bioaccumulative (vPvB). REACH and RoHS directives already restrict legacy compounds like polybrominated diphenyl ethers (PBDEs).
Market reality: The global bromine FR market was valued at USD 2.47 billion in 2025, with electrical and electronics representing USD 1.13 billion (46% of total demand). Despite regulatory scrutiny, brominated systems remain widely used where performance and cost constraints demand them, particularly in TBBPA-based FR-4 circuit boards, which represent over 90% of global PCB production.
Phosphorus-Based Flame Retardants
Phosphorus FRs span both gas-phase and condensed-phase mechanisms depending on chemical structure:
Gas-phase acting:
- Organophosphates and phosphinates (resorcinol diphosphate, aluminum/zinc phosphinate salts)
- Used in electronic plastics and engineering polymers
- Aluminum phosphinate (Clariant Exolit OP) received GreenScreen Benchmark 3 rating for PA6 and PP applications
Condensed-phase acting:
- Ammonium polyphosphate (APP): promotes char formation; central to intumescent fire-protection coatings
- DOPO (9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide): reactive phosphorus FR used in circuit board epoxy resins
Why formulators choose phosphorus FRs:
- Effective at lower loadings than mineral fillers
- No halogen-related regulatory concerns
- Compatible with many engineering polymers
Formulation watch-outs:
- Can increase smoke output
- Not universally effective across all polymer types
- Some organic phosphates face emerging regulatory scrutiny (TCEP banned under REACH Annex XIV as of August 2015; triphenyl phosphate added to SVHC list November 2024)
Mineral Filler Flame Retardants
Aluminum Trihydrate (ATH):
- Decomposes at 180-200°C, releasing water vapor
- Suitable for polymers processed below 200°C
- Complete decomposition by 300°C
- Endothermic absorption cools polymer surface
Magnesium Hydroxide (MDH):
- Decomposes at 330°C
- Thermally stable up to 110°C higher than ATH
- Enables processing of engineering thermoplastics and polyamides
- Preferred for LSZH wire and cable jacketing
Performance characteristics:
- Require 50-60 wt% loading in polyolefins to achieve meaningful FR performance
- High loadings degrade tensile strength and elongation
- Surface-treated grades and compatibilizers mitigate mechanical property loss but add cost
- Clean combustion profile: only water vapor and metal oxides, no toxic or corrosive gases
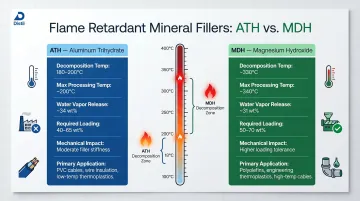
The temperature boundary between ATH and MDH creates a hard processing constraint: if your polymer requires extrusion above 200°C, MDH is the only viable mineral filler option.
Intumescent Systems
Intumescent FRs rely on three-component chemistry:
- Acid source — ammonium polyphosphate (APP)
- Carbon source — pentaerythritol (PER) or the polymer backbone
- Blowing agent — melamine
During fire exposure, the acid catalyzes dehydration of the carbon source into char. Simultaneously, the blowing agent releases non-flammable gases (NH₃, N₂), expanding the char into a thick, insulating foam barrier. This structure physically protects the substrate from heat and blocks fuel gas release.
Applications:
- Building and construction firewall barriers
- Fire-resistant coatings and paints (ASTM E119, ASTM E2768)
- Aerospace and mass transport components
- Wire and cable in critical infrastructure
Challenges:
- Moisture sensitivity
- Limited processing temperature windows
- Durability under sustained flame exposure varies by formulation
Inorganic Synergists and Nanocomposites
None of the primary FR chemistries above operates in isolation. Inorganic synergists and nanocomposites extend their performance without increasing loadings, addressing specific gaps that primary systems leave behind:
- Zinc borate: suppresses afterglow in mineral-filled compounds; used at 3-25 phr in halogen systems, 10-250 phr in halogen-free systems; releases water of hydration above 290°C
- Tin compounds (zinc hydroxystannate): reduce smoke in halogenated systems
- Silicon-based materials: contribute to protective oxide barrier formation
Typical loadings: 1-5 wt% to address specific performance gaps.
Polymer nanocomposites (organically treated layered silicates/clays, carbon nanotubes) form a clay- or fiber-rich char network during burning:
- At 1-10 wt% loading, dispersed nanoparticles reduce peak HRR by up to 50%
- Polystyrene/montmorillonite nanocomposite reduced PHRR from 1,025 kW/m² to 520 kW/m²
- Carbon nanotubes achieve approximately 50% PHRR reduction at only 1 wt% loading
Critical limitation: Nanocomposites alone do not meet standard fire tests (UL 94). They function most effectively as universal FR synergists — when combined with primary FR chemistries, the nanocomposite structure amplifies performance and can reduce the required loading of the primary FR by 20-30%.
Additive vs. Reactive Flame Retardants
Additive FR systems are physically blended into the polymer matrix during compounding. They dominate the market because they are:
- Cost-effective and compatible with a broad range of polymers
- Easy to incorporate without modifying the base synthesis route
- Flexible to adjust across formulation iterations
These advantages come with trade-offs worth accounting for at the design stage:
- Blooming — surface migration over time, especially at higher loadings
- Leaching or extraction during service
- Loss of FR activity during recycling
- Mechanical property degradation at high loadings
Where additive systems fall short on durability, reactive FR systems offer a fundamentally different approach: the flame retardant is covalently bonded into the polymer backbone during synthesis itself. TBBPA in epoxy circuit boards is the canonical example — reacted into the epoxy backbone via chain extension, it achieves 18–21% bromine by weight in more than 90% of FR-4 boards produced globally. Once chemically bound, TBBPA no longer behaves as a discrete molecule, eliminating migration risk entirely.

Reactive systems offer distinct performance advantages:
- Permanent integration — no blooming, leaching, or recycling-related FR loss
- Better-balanced mechanical, thermal, and flammability properties across the service life
- More consistent performance over the full product lifecycle
The trade-offs are primarily on the development side:
- Requires modified synthesis routes and longer development timelines
- Higher upfront cost and reduced formulation flexibility once the backbone is set
Regulatory tightening around certain additive-type halogens and plasticizers is accelerating interest in reactive FR chemistries and halogen-free polymer systems. The right choice depends on regulatory outlook, recyclability requirements, and total formulation cost over the product lifecycle.
Key Applications Across Industries
Electronics and Electrical
Circuit boards (FR-4 epoxy laminates), connector housings, cable insulation, and device enclosures must comply with UL 94 V-0 and IEC 60695 standards. UL 94 V-0 is the most stringent vertical burn classification, requiring:
- Individual flame time ≤10 seconds
- Total flame time ≤50 seconds for five specimens
- No flaming drips igniting cotton indicator
Dominant chemistries:
- TBBPA (reactive) in epoxy laminates
- Phosphorus FRs (DOPO, phosphinate salts) in halogen-free electronics
- ATH in lower-temperature applications
The halogen-free FR market was valued at USD 6.4 billion in 2024, projected to reach USD 10.5 billion by 2030 — driven by REACH/RoHS compliance requirements and OEM procurement policies shifting toward halogen-free chemistries.
Wire, Cable, and Construction
Infrastructure applications demand a different compliance profile than electronics. Low-smoke zero-halogen (LSZH) cables in tunnels, data centers, and public buildings use ATH and MDH mineral filler systems to meet IEC 60332 and EN 50267 standards. Building insulation (polyurethane and polystyrene foams) relies on intumescent and phosphorus systems.
Regulatory drivers shaping this segment:
- EU Construction Products Regulation (EN 50575)
- NFPA codes in North America
- IEC 60332 for flame propagation
- EN 50267 for acid gas emission
Meeting these standards requires more than flame suppression — smoke toxicity becomes the critical selection variable. Enclosed spaces penalize incomplete combustion products, making condensed-phase FRs (mineral fillers, intumescent systems) preferred over gas-phase inhibitors.
Automotive and Industrial Packaging
Automotive interior components (instrument panels, seat foams, wire harnesses) must pass FMVSS 302 and OEM-specific fire tests. FMVSS 302 measures horizontal burn rate, requiring:
- Burn rate ≤102 mm/min, or
- Self-extinguishment within 60 seconds/51 mm
Common solutions:
- Phosphorus FRs in polypropylene compounds
- Intumescent systems in polyurethane seat foams
- MDH in wire harnesses and under-hood components
Distil works with automotive and wire and cable manufacturers on FR polymer compound development — from initial formulation trials through commercial-scale production. Using a network of 20+ manufacturing partners across India, Distil handles compound sourcing, formulation tuning, and scale-up execution as a single point of contact.

Choosing the Right Flame Retardant System
Selection Framework
Step 1: Host polymer and processing temperature
The polymer's chemistry and processing temperature set the first constraint. If your compound requires extrusion above 200°C, ATH is eliminated — MDH becomes the only viable mineral filler option. Polyamides, engineering thermoplastics, and high-temperature polyolefins require MDH or phosphorus-based systems.
Step 2: Required fire test standard
The fire test standard defines the performance target:
- UL 94 V-0: Gas-phase FRs (halogenated, phosphorus) excel
- Cone calorimeter (ISO 5660-1): Char-forming systems (intumescent, nanocomposites) reduce peak HRR
- IEC 60332 / EN 50267 (LSZH): Mineral fillers dominate due to low smoke/toxicity profile
Step 3: Application environment
Outdoor UV exposure, food contact, or medical device use impose additional restrictions:
- UV exposure requires stabilizers compatible with the FR system
- Food contact limits leachable additives, favoring reactive FRs
- Medical applications often prohibit halogenated compounds
Regulatory and Sustainability Factors
Halogen-free demand is accelerating:
The halogen-free FR market (USD 6.4 billion, 2024) already exceeds the bromine FR market (USD 2.47 billion, 2025) by a factor of 2.6×. Drivers include:
- EU REACH restrictions on aromatic brominated FRs
- RoHS limitations on PBB and PBDE in electronics
- End-of-life recyclability concerns
- OEM procurement policies prioritizing halogen-free chemistries
The environmental and toxicological profile of the FR additive — not just its fire performance — is now a procurement criterion for many brand owners.
Formulation Complexity
FR additive selection is rarely a single-component decision. Synergist combinations, compatibilizers, and processing stabilizers must be co-optimized. For example:
- Halogenated systems require Sb₂O₃ at validated ratios (1:2 to 1:3.5)
- Mineral filler systems need surface treatments and compatibilizers to preserve mechanical properties
- Nanocomposite synergists enable 20-30% reduction in primary FR loading
For teams without deep FR formulation expertise in-house, partnering with an R&D-led specialty chemicals platform like Distil can cut development timelines and reduce formulation risk. Distil's process covers each stage from application brief to commercial production:
- Define requirements — map processing conditions, performance targets, and regulatory constraints upfront
- Design the formulation — a team with 45+ years combined experience from Dow, BASF, L'Oréal, Huntsman, and Reliance Industries develops bespoke FR systems
- Validate in the lab — trials confirm batch-to-batch consistency in processing and end-use performance
- Optimize for manufacturing — formulations are aligned to partner facility conditions before scale-up begins
- Scale without capex — production runs through 20+ vetted partner facilities under unified quality control, eliminating process drift
A single point of contact manages the full journey, from lab to commercial volume.
Frequently Asked Questions
What are flame retardant polymer additives?
Flame retardant polymer additives are chemical compounds blended into or reacted with polymer matrices to interrupt the combustion cycle. They prevent ignition, slow flame spread, or reduce heat release, enabling plastics to meet fire safety standards across industries.
What flame-retardant treatments are used for plastics?
The three main approaches are: incorporating FR additive packages (halogenated, phosphorus, mineral fillers, intumescent) during compounding; applying surface coatings or intumescent paints; and building reactive FR chemistry into the polymer backbone during synthesis.
What flame-retardant additives are used in HDPE?
HDPE processed above 200°C typically uses magnesium hydroxide (MDH) as the primary mineral filler FR at 50–60 wt% loading. Formulators may combine MDH with intumescent additives or nanocomposite synergists to reduce total loading while meeting LSZH cable or pipe standards.
What are common flame-retardant polymers?
Inherently flame-retardant polymers include PVC, PTFE, polycarbonate, and PPS, which have low flammability due to their chemical structure. Commodity polymers like polyolefins, polystyrene, and polyurethane require FR additive treatment to meet fire safety standards.
What is the difference between additive and reactive flame retardants?
Additive FRs are physically blended into the polymer and can migrate or leach over time. Reactive FRs are chemically bonded into the polymer chain during synthesis, offering more durable performance and better property retention but at higher development cost.
Are halogen-free flame retardants as effective as brominated systems?
Halogen-free alternatives (phosphorus, mineral filler, intumescent) can match brominated FR performance in many applications, particularly wire/cable and construction, though higher loadings or synergist combinations are often needed. The performance gap has narrowed considerably as halogen-free chemistry has matured under regulatory pressure.



